 
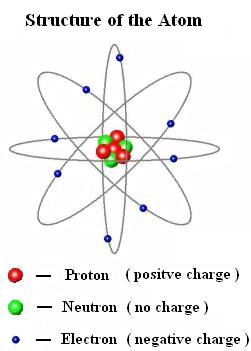
Chemical reactions largely involve atoms or groups of atoms and the interactions between their electrons. In general, electrons are easier to add or remove from an atom than a proton or neutron. The particles within an atom are bound together by powerful forces.Isotopes and ions of an atom with a constant number of protons are all variations of a single element. Varying the number of electrons results in ions. Varying the number of neutrons results in isotopes. Thomsons plum pudding model of the atom had negatively-charged electrons embedded within a positively-charged 'soup. The number of protons (also known as its atomic number) determines the element.Almost all of the mass of an atom is in its nucleus almost all of the volume of an atom is occupied by electrons.The average size of an atom is about 100 picometers or one ten-billionth of a meter. Electrons can also move between orbitals. Those that lose electrons become positively charged, and those that gain electrons become negatively charged. Technically, an electron can be found anywhere within the atom, but spends most of its time in the region described by an orbital. Some electron shells resemble spheres, but others look more like dumb bells or other shapes. Simple models show electrons orbiting the nuclear in a near-circular orbit, like planets orbiting a star, but real behavior is much more complex. Electrons are organized into shells, which is a region where an electron is most likely found. Electrons move around outside the nucleus.The nucleus carries a positive electrical charge. The nucleus of an atom contains protons and neutrons. 
The mass of a proton is 1840 times greater than the mass of an electron. The mass of a proton is essentially the same as that of a neutron.
0 Comments
Leave a Reply. |




 RSS Feed
RSS Feed
